Dear RWE4Decisions Learning Network,
Welcome to the Spring 2026 RWE4Decisions Newsletter! We recommend to ‘Download external images’ for the full experience (find this button at the top of the email).
This edition focuses on key activities from the second half of 2025 and the first quarter of 2026, including:
two Roundtables focused on RWE in Oncology and RWD collection in Alzheimer’s disease
two Public Webinars on transforming RWE with AI, and on the use of registries to improve rare disease RWD
the 2025 Annual Symposium, exploring how RWD can meaningfully support decision-making for HTA bodies in light of the evolving EU health data landscape
Following a very busy beginning to 2026, we also look forward to our upcoming plans — keep reading to find out more about our next events, the Stakeholder Actions Focus Groups, our ongoing papers, our participation in the 2026 HTAi Annual Meeting, and the latest changes to the RWE4Decisions Steering Group.
We hope you’ll enjoy the read!
P.S. We are quite active on LinkedIn – click here to follow the RWE4Decisions page and not miss any updates.
On 18 September 2025, a multistakeholder invitation-only Roundtable examined the evolving role of RWE in HTA decision-making in oncology. The discussion reinforced the important role of RWE in bridging evidence gaps and supporting patient access to innovative therapies.
Dr Beate Wieseler (IQWiG) set the stage with a keynote address on the HTA Coordination Group’s guidance on the validity of clinical studies and on evidence synthesis. This was followed by presentations of practical oncology HTA case studies from various jurisdictions, and by rich breakout room discussions on data quality and transferability challenges, the role of registries, and the need for reference cases to guide the generation of RWE.
These discussions contribute to the ongoing efforts to improve how RWE is generated and used in HTA decision-making across oncology.
The key meeting takeaways are being included in a peer-reviewed journal paper; stay tuned to find out more.
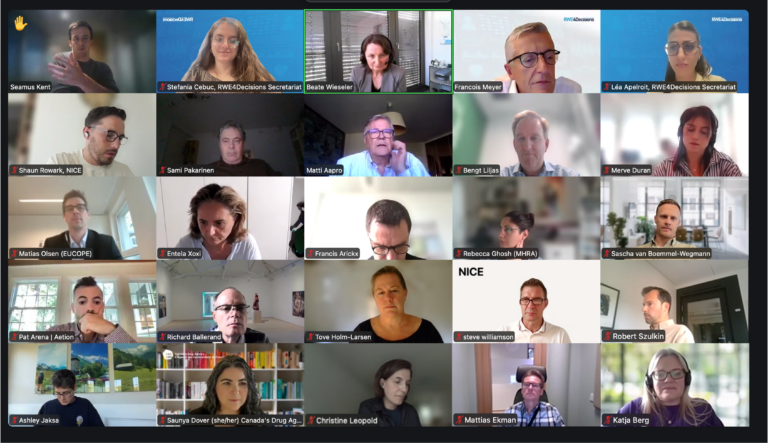
On 15 October 2025, a public Webinar brought together HTA bodies, patient representatives, and industry to examine how AI is being used in evidence generation, data analysis, and decision-making across the RWE lifecycle.
Speakers discussed the opportunities and challenges of integrating AI into evidence generation, emphasising the importance of human oversight, trust, and collaboration.
The discussion highlighted practical applications of AI from different stakeholders’ perspectives. It revealed differing levels of adoption across organisations as well as common implementation challenges. Participants also emphasised the need for shared frameworks to support reliable, transparent, and ethically sound use of AI in generating RWE.


Over 80 participants joined us for the online edition of the Annual RWE4Decisions Symposium on 20 November 2025. The event focused on how RWD can meaningfully support decision-making in the context of the HTA Regulation, the European Health Data Space, the rise of AI in evidence generation, and the future of HTA collaboration across Europe. Speakers included representatives from the Danish EU Presidency, European Commission, European Medicines Agency, national HTA/Payers, and patient and industry associations.
Key takeaways include:
RWE has the potential to reduce knowledge gaps at key decision points such as pricing negotiation and reimbursement. Moreover, ongoing initiatives like DARWIN-EU and EHDS are vital to improving data quality and availability.
Future success depends on coordinated access, data standardisation, and collaborative models across countries.
Better data makes for better decisions and better lives.
A meeting report will be coming out before the summer – stay tuned!
The 2026 work programme started off on 18 February, with an invitation-only Roundtable on Alzheimer’s Disease.
We welcomed more than 60 attendees to discuss some of the key challenges facing the collection of RWD to support HTA and payer decisions.
While Alzheimer’s disease was used as a case study, the discussion made it clear that many of the principles discussed are relevant across disease areas.
Key insights reveal that:
Patients’ voices are crucial: they want to contribute information that is simple, not overly burdensome, and flexible in frequency,
Defining the core datasets is essential to understanding missing information from existing data sources,
It is important to consider not only clinical outcomes but also societal values relevant for HTA payer decision-making and,
Uncertainty will always exist, and must be balanced against unmet need and the particular population.
On 8 April 2026, over 180 participants joined our Public Webinar to discuss how HTA bodies, clinicians, patient organisations, and industry can work together to improve the availability and quality of Rare Disease RWD to support HTA/Payer decision-making through the use of registries.
Our keynote speaker, Prof. Maurizio Scarpa, MD, PhD (MetabERN), presented an insightful keynote address on approaches to improving data generation and use across stakeholders to deliver value in rare diseases, including through the development of registries in ERNs and the 2025 HLM for Rare Diseases. This was followed by a lively panel discussion on the use of RWD across the lifecycle of Rare Disease medicine development, patient involvement for RWD collection, and the role of RWD in HTA decision-making.
The slides and recording are already available, and a meeting report will be coming out soon – stay tuned!

On 3 June, a Roundtable on the use of RWE and patient-relevant outcomes will take place via Zoom.
Roundtables are invitation-only meetings; please get in contact with the Secretariat if you would like to attend.
Join us on 15 July for a Webinar co-moderated by Matias Olsen (EUCOPE) and François Meyer (RWE4Decisions) to discuss current practices in horizon scanning, early dialogues, and what changes could be made to improve consideration of RWE generation throughout the drug development lifecycle.
More information on the speaker lineup and a registration link will follow soon — keep an eye out for our emails!

DON’T MISS OUT
RWE4Decisions will be at the HTAi Annual Meeting (6-10 June 2026, Istanbul, Turkey)
RWE4Decisions will be joining the HTAi 2026 Annual Meeting and hosting a panel entitled “AI Transforming Evidence Generation for HTA”. It will be moderated by Karen Facey (RWE4Decisions) and joined by Stephen Duffield (NICE), Jing Wang-Silvanto (Astellas), and Andrew Spiegel (Global Colon Cancer Association) as the presenters.
SAVE THE DATE
The Annual RWE4Decisions Symposium will be taking place in person on 23 September, from 09:00 to 14:00 CEST.
Join us to discuss more about optimising RWE to deliver value in healthcare!
More updates will follow soon — stay tuned!
RWE4Decisions, alongside volunteering authors, is in the process of developing and publishing three scientific papers. These build on insights from our previous Webinars and Roundtables focused on Transportability, Oncology, and AI.
We look forward to sharing more as these papers develop — until then, make sure to check out our previous publications by clicking the button below.
In 2024, RWE4Decisions launched the second iteration of our Stakeholder Actions to Generate Better RWE for HTA/Payers, followed by a journal publication in 2025.
This year, we want to explore how the actions are being implemented in practice. For this purpose, RWE4Decisions has been organising Stakeholder Action Focus Groups to discuss which policy developments have shaped the health data landscape in the past 18 months, what ongoing projects and initiatives can relate to the actions, and what learnings can be drawn for better implementation.
The learnings and stories from these Groups will be shared on our website later in the year. Stay tuned!
Do you have any experiences or stories that relate to the Stakeholder Actions? We’d be grateful to hear about it! Please submit your implementation story on the RWE4Decisions Website or write us an email with more information.
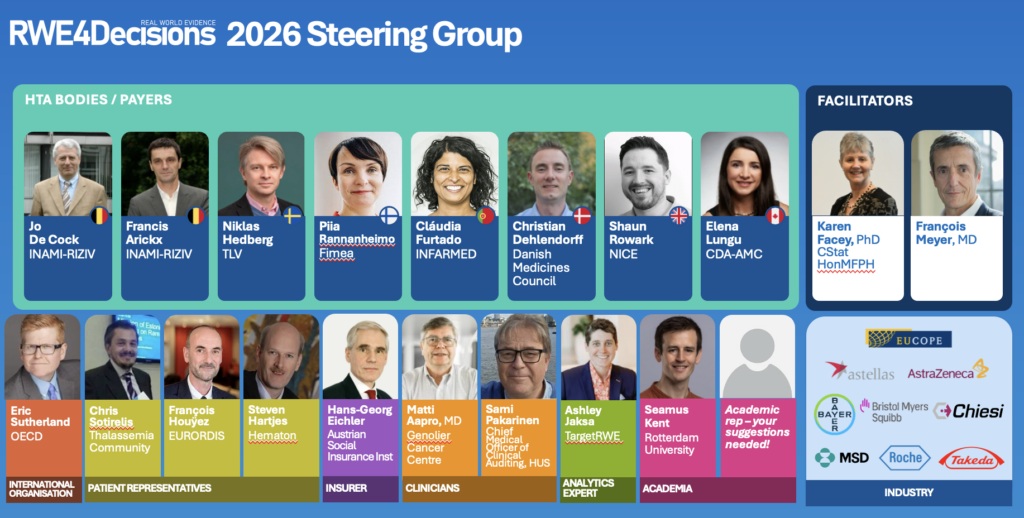
Over the last couple of months, we bid our farewells to a number of Steering Group members who have been closely involved in shaping the work of RWE4Decisions over the past years: Francis Arickx (INAMI-RIZIV), Elena Lungu (CDA-AMC), Entela Xoxi (Universita Cattolica Sacro Cuore), and Julien Delaye (EURORDIS). We heartfully thank them for their contributions and dedication.
At the same time, we happily welcome a number of new members, bringing new ideas that will guide our work going forward: Dr. Sami Pakarinen (HUS), François Houÿez (EURORDIS), Steven Hartjes (Hematon), and Victoria Hodgkinson (University of Calgary).
At the end of 2025, we bid our heartfelt farewell to Alina Pavel, whose dedication across the past years has shaped the RWE4Decisions Secretariat’s work.
The Secretariat has been happy to welcome two new members: Emma Loebler, who we thank for her hard work and dedication since joining the team in October 2025. Her ample experience in healthcare consultancy has paved the way for valuable outputs. In addition, newly-joined Ysaline Van Malderen brings in fresh insights and a background in digital health.
|